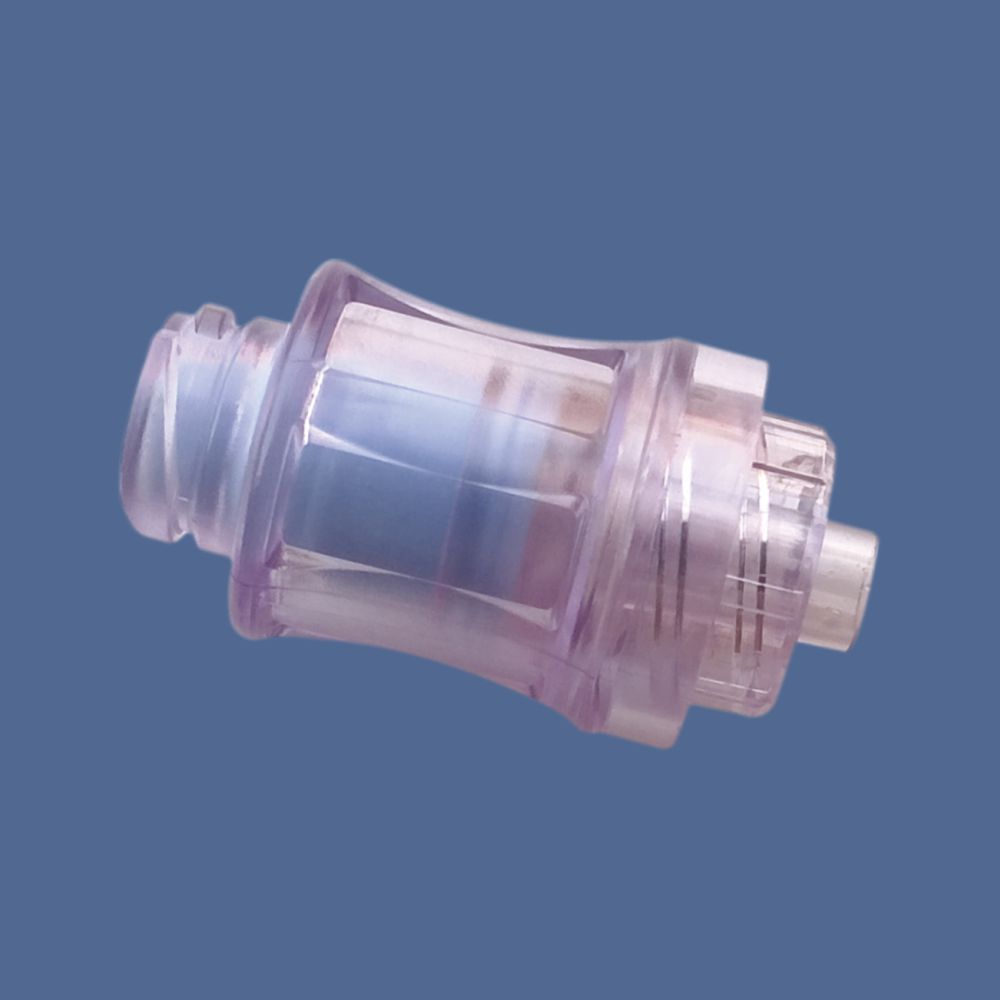
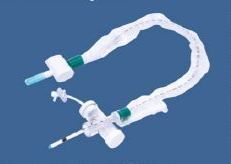
Smart K-Site, Ref. BI 2000P, Two way PU Extension with two Needle Free Vlave
Price 95.0 INR/ Piece
Smart K-Site, Ref. BI 2000P, Two way PU Extension with two Needle Free Vlave Specification
- Storage Instructions
- Store in a cool, dry place, away from direct sunlight
- Accuracy
- High flow accuracy for medical use
- Function
- Provides safe IV access and medication administration with minimized infection risk
- Shelf Life
- 3-5 years (unopened pack)
- Instruments Type
- IV Accessory/Extension Set
- Features
- Two needle-free valves, kink resistant tube, leak-proof design, latex-free, luer lock connectors, pressure resistant
- Usage Type
- Single use, Disposable, Sterile
- Equipment Type
- Two-way Polyurethane (PU) Extension Set with Needle Free Valves
- Material
- Medical grade Polyurethane (PU), DEHP-free, Non-toxic
- Condition
- New
- Technology
- Needle Free Valve Technology
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- Yes
- Noise Level
- Silent/Non-electric
- Operating Type
- Manual
- Use
- Intravenous Infusion Extension and Drug Delivery
- Dimension (L*W*H)
- Approx. 200 mm (length)
- Weight
- Lightweight (approx. 20g)
- Color
- Transparent with colored coded hubs (Clear/Blue/White)
- Tube Type
- Kink-resistant, pressure tolerant up to 300 psi
- Valve Type
- Non-return/anti-reflux needleless connectors
- Connector Type
- Male luer lock at one end, Female luer lock at opposite end with two needle-free access ports
- Packaging
- Individually packed, sterile, peel pouch
- Compliance
- Conforms to ISO 8536-9 and relevant IV therapy standards
- Priming Volume
- Approx. 2.0 ml
- Latex Status
- Latex-free, DEHP-free
- Sterilization Method
- Sterile, Ethylene Oxide (EtO) sterilized
- Compatibility
- Universal with standard IV therapy components and infusion sets
Smart K-Site, Ref. BI 2000P, Two way PU Extension with two Needle Free Vlave Trade Information
- Minimum Order Quantity
- 100 Pieces
- Supply Ability
- 500 Pieces Per Month
- Delivery Time
- 1 Week
About Smart K-Site, Ref. BI 2000P, Two way PU Extension with two Needle Free Vlave
- Design: Dual-port (Two-Way) extension set with needle-free access.
- Material: Polyurethane (PU) tubing, which is flexible and kink-resistant.
- Components: Two needle-free valves with silicone sealing membranes for automatic sealing.
- Safety: DEHP-free and Latex-free, making it suitable for sensitive patient populations.
- Dimensions & Flow: Length is typically (or), with abore and a flow rate of.
- Usage: Used for multi-access, simultaneous, or alternate IV therapy to reduce infection risk.
Advanced Needle-Free Technology
Smart K-Site integrates non-return needleless connectors to enable safe IV access and medication delivery without needles. This technology not only reduces needlestick injury risks but also ensures excellent compatibility with existing IV therapy and infusion sets, making clinical workflow safer and more efficient.
High Performance and Safety Standards
Engineered from medical-grade polyurethane, Smart K-Site offers kink resistance, reliable pressure tolerance up to 300 psi, and high flow accuracy. It fully conforms to international medical standards such as ISO 8536-9, ensuring consistent performance and utmost patient safety in intravenous therapy applications.
User-Friendly and Convenient Design
With its clear, color-coded hubs and lightweight, portable build, Smart K-Site is designed for ease of use, rapid identification, and hassle-free handling. The individually packed, sterile peel pouch ensures product integrity and single-use disposability, contributing to stringent infection control in healthcare environments.
FAQ's of Smart K-Site, Ref. BI 2000P, Two way PU Extension with two Needle Free Vlave:
Q: How is the Smart K-Site, Ref. BI 2000P, used during intravenous therapy?
A: Smart K-Site is connected using the male luer lock at one end and female luer lock at the opposite end, acting as an extension to standard IV lines. Its needle-free valves allow safe administration of fluids or medications and enable access without needles, reducing the risk of infection and accidental needlesticks.Q: What makes the Smart K-Site suitable for high-pressure infusion applications?
A: Constructed from pressure-tolerant, medical-grade polyurethane, the Smart K-Site can withstand pressures up to 300 psi. Its kink-resistant tubing ensures unimpeded fluid flow, making it ideal for demanding medical environments where reliability is essential.Q: When should the Smart K-Site extension set be replaced?
A: Smart K-Site is a single-use, disposable device and should be discarded immediately after one patient use to maintain sterility and prevent cross-contamination. Do not attempt to reuse or re-sterilize the extension set.Q: Where can the Smart K-Site be stored before use?
A: Store the device in a cool, dry location away from direct sunlight to preserve its sterility and extend its shelf life, which ranges from 3 to 5 years for unopened packs.Q: What are the main benefits of needle-free valve technology in Smart K-Site?
A: The needle-free valves minimize the risk of bloodstream infections and eliminate needlestick injuries. They also streamline the IV access process, making it safer and faster for both clinicians and patients.Q: How does the Smart K-Site ensure compatibility with standard IV therapy equipment?
A: Smart K-Site features universal male and female luer lock connectors, designed to fit all standard IV sets and infusion components, ensuring seamless integration with existing hospital equipment.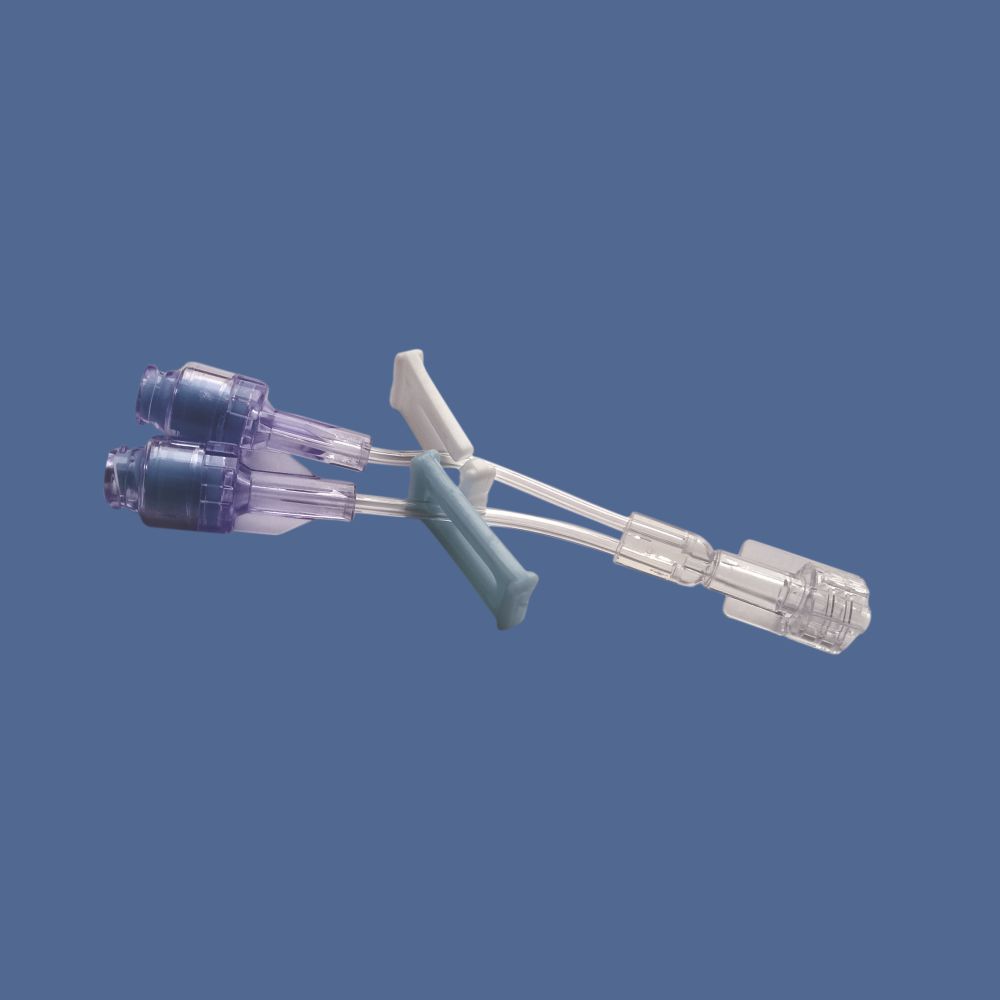

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Medical Supplies Category
Dialo K-Site, Ref- DKS 100
Price 50.0 INR / Piece
Minimum Order Quantity : 100 Pieces
Material : Other, Stainless Steel and ABS Plastic
Condition : New
Equipment Type : Dialyzer Reprocessing Unit
Operating Type : Automatic
Closed Suction System
Minimum Order Quantity : 5 Pieces
Material : Other, Medical Grade PVC
Condition : New
Equipment Type : Closed Suction System
Operating Type : Manual
Foley Catheter
Price 22 INR / Piece
Minimum Order Quantity : 200 Pieces
Material : Latex
Condition : New
Equipment Type : Foley Balloon Catheter siliconized
Uro Meter
Price 70 INR / Piece
Minimum Order Quantity : 250 Pieces
Material : Plastic
Condition : New
Equipment Type : Uro Meter
Operating Type : Manual

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry